A New and Improved, Safer Alternative to Imatinib
At Inhibikase, we developed IkT-001Pro as an outgrowth of our efforts to improve the safety of the first FDA-approved Abl kinase inhibitor, imatinib (marketed as Gleevec®), which is commonly taken for hematological cancers that arise from Abl kinase mutations in the bone marrow or for gastrointestinal cancers that arise from c-Kit or PDGFRa/b mutations in the stomach.
IkT-001Pro has shown to be potentially as much as three times safer than imatinib in non-human primates, blunting the severe gastrointestinal side effects of imatinib therapy that occur following oral administration. Removing these gastrointestinal side effects has the potential to significantly improve the number of patients that reach and sustain complete cytogenetic response in stable-phase chronic myelogenous leukemia (CML). We believe that removing these side effects will also improve patient adherence to daily therapy and improve patients' quality of life while on therapy.
IkT-001Pro has received Orphan Drug Designation for stable-phase CML and will follow the development pathway for approval through the 505(b)(2) regulation. This pathway would allow us to rely, in part, on data in the public domain or the FDA’s prior conclusions regarding the safety and effectiveness of an approved compound.
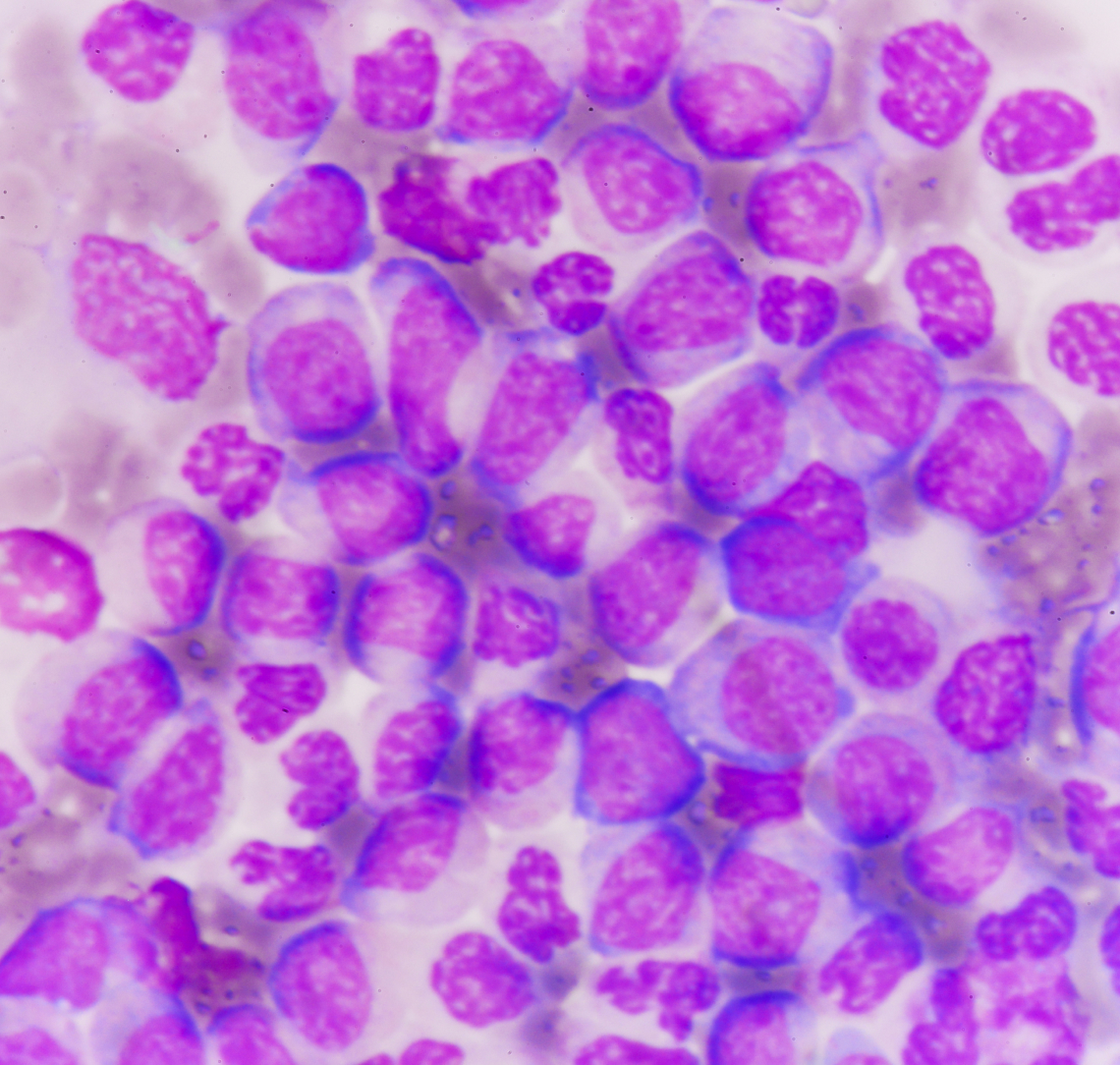
About Chronic Myeloid Leukemia
Chronic myeloid leukemia (CML)1 is a slowly progressing cancer that affects the blood and bone marrow. In CML, a genetic change takes place in immature myeloid cells—the cells that make most types of white blood cells. This change creates an abnormal gene product, called BCR-ABL, which transforms the cell into a CML cell. Leukemia cells increasingly grow and divide in an unregulated manner, eventually spilling out of the bone marrow and circulating in the body via the bloodstream. Because they are not fully mature, CML cells are unable to work properly to fight infections. In time, the cells can also settle in other parts of the body, including the spleen. CML is a form of slow-growing leukemia that can change into a fast-growing form of acute leukemia that is difficult to treat.
1Also known as chronic myelogenous leukemia, chronic myelocytic leukemia, and chronic granulocytic leukemia (CGL)